Orthosilicic acid exists in drinking water. This is an idealised presentation because of polymerisation reactions. In practice, analysts measure “reactive silica.”
Well – that’s what we can infer from a new campaign of the Fluoride Action Network (FAN).
But what is the link with fluoride – the whole reason for FAN’s existence? Well, they base this campaign on the well-known hydrolysis of the fluorosilicates used in community water fluoridation to form the hydrated fluoride anion and silica. (Although these campaigners are confused here as they will also often claim fluorosilicates do not hydrolyse and survive to come out of your tap and poison you).
Then they claim that “silicic acid” (silica in solution) dissolves lead from the pipes and fittings and this lead causes brain damage. A double-barreled danger as they also claim the fluoride also damages your brain. What’s more – they also claim that “silicic acid” may dissolve your teeth!
But there are two problems with this:
- Your drinking water contains silica whether it is fluoridated or not. So their warnings about the silica in fluoridated water should also be valid for “fluoride-free” water which they promote!
- They do not have a viable chemical mechanism for silica dissolving your pipes (and there are plenty of other mechanisms which can result in corrosion of pipes anyway). The same for your teeth. This claim is just not supported by the chemical literature.
I will just concentrate here on the “evils” of silica (or “orthosilicic acid”) that are being promoted by FAN and leave the lead story for another day. These “evils” all come down to concepts being promoted by Richard Sauerheber who FAN describes as “the ultimate citizen chemist.” (OK, he is their ultimate citizen chemist). His argument is presented in Silicic Acid – How Does Fluorosilicic Acid Leach Lead? Why Does Fluorosilicic Acid Leach Lead So Much More Than Sodium Fluoride?
Is silicic acid the bogy Sauerheber claims?
His article is confused and convoluted. But it starts with the assumption that silica in drinking water (silicic acid or orthosilicic acid) is bad. He declares:
“Neither fluoride nor silicic acid are constituents of normal pristine human or mammalian blood, but rather are contaminant materials, . . “
And he states:
“The mass treatment of public fresh drinking water with industrial fluorosilicic acid to produce fluoride ion at 1.0 ppm also produces approximately 6 ppm sodium ion and .7 ppm orthosilicic acid. None of these is found in or belongs in fresh drinking water.”
Let’s stop right there and check out his claim that sodium and “orthosilicic acid” are not found in fresh drinking water.
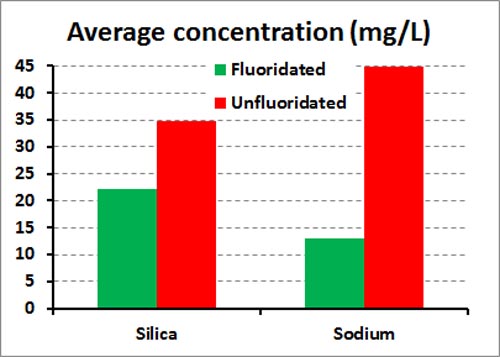
Here is some data for drinking water in fluoridated and unfluoridated areas of Auckland, New Zealand, taken from the WATERCARE ANNUAL WATER QUALITY REPORT 2013. The data are for 15 unfluoridated treatment plants and 8 fluoridated treatment plants.
These are average figures over all the plants. But values for silica as high as 64 mg/L in the unfluoridated plants and 44 mg/L in the fluoridated plant were recorded. The corresponding figures for sodium were 140 and 22 mg/L.
So Sauerheber is completely wrong. Unfluoridated water does contain silica and sodium. And at concentrations much higher than could be accounted for by added fluoridating chemicals – he calculated 0.7 mg/L – 0.7 ppm) for silica. In fact, the values are higher for the unfluoridated treatment plants in these examples.
Fluoridation makes a minuscule contribution to the concentration of these chemical species in drinking water.
Forget about fluoridation. If silica (and sodium) are such problems then Sauerheber should be campaigning against unfluoridated water as well. Even the “pristine” water in his local river or spring – silica is a normal and natural component of surface and bore waters.
But Sauerheber is also wrong about the dangers of silica in drinking water. Of course silica is present in “normal pristine human or mammalian blood” because it is part of our diet (Bisse et al 2005). It is a component of many of our foods. Sauerheber is simply attempting to confuse the issue because of the current lack of knowledge about the role of silica in the body.
But Jugdaohsingh et al., (2015) say:
“Silicon (Si) is a natural trace element of the mammalian diet and although it has not been demonstrated unequivocally that mammals have a requirement for Si there is increasing evidence to suggest that it may be important for the normal health of bone and the connective tissues. Indeed, severe dietary Si deprivation in growing animals appears to cause abnormal growth and defects of the connective tissues”
Given that silica appears to be important the presence of it in our drinking water is as an advantage. According to Jugdaohsingh (2007):
“Drinking water and other fluids provides the most readily bioavailable source of Si in the diet, since silicon is principally present as Si(OH)4, and fluid ingestion can account for ≥ 20% of the total dietary intake of Si.”
One trick Sauerheber uses is to cite reports of the danger of inhaled silica dust – which can cause cancers and silicosis. Completely irrelevant because of its different chemical form. This is equivalent to the trick often used by anti-fluoride campaigners of citing reports of results of industrial pollution or studies from areas of endemic fluorosis to support their attacks on community water fluoridation.
Will silica dissolve your teeth?
This claim is completely unsupported – no citations and purely a figment of Sauerheber’s imagination. He claims:
“Orthosilicic ‘weak’ acid has been long used in agriculture to break down solid calcium phosphate Ca3(PO4)2, thereby releasing soluble phosphate ion in soils even at neutral pH, for uptake by plant life. The reaction of silicic acid with calcium phosphate under neutral pH conditions is:
H4SiO4 + Ca3(PO4)2 → HPO4-2 + 3Ca2+ + PO4-3+ H3SiO4–.
This reaction occurs at a pH where any strong acid would have been neutralized. Orthosilicic acid is reluctant to dissociate and can break down calcium phosphate. This reaction is relevant not only to calcium phosphate in soil but also to calcium phosphate in teeth enamel. By means of orthosilicic acid, enamel is subject to slow and progressive degradation. “
This is a new one on me – and I spent many years researching the dissolution of apatite (the natural calcium phosphate) in soil and the factors influencing that. This arose because unacidulated phosphate rocks were being used in New Zealand agriculture. These material contain insoluble phosphate – in contrast to superphosphate which contain soluble phosphate.
The phosphate rocks used are mainly apatites and are complex (because of isomorphous substitution). A specific chemical equation for their dissolution depends on composition and environmental pH. But, in general, acid (H+) reacts with the apatite to produce Ca2++ H2PO4–, H2O, F–, Cl–, CO2, etc.
H+ + Ca10(PO4)6(OH,F,Cl)2 → Ca2+ + H2PO4– + H2O + Cl– + F– + CO2
Dissolution is promoted by the presence of acid (H+) and removal of dissolution products (particularly Ca2+). The calcium in soil solution can inhibit apatite dissolution – it drives the equation above to the left. The later is important because New Zealand agricultural soils have relatively high levels of calcium. On the other hand, our research showed that when soils are leached to remove calcium this can promote dissolution of the natural fluorapatite in the soil. (Perrott and Kear 2004). Removal of calcium from solution drives the above equation to the right.
Apatite particle size, fluoride content and substitution of other species in the apatite structure can also influence the dissolution rate of these materials in soil. But silica, or silica in soil solution – that is a new one on me!
Pity Sauerheber didn’t give a citation to support his claim that silica “has been long used in agriculture to break down solid calcium phosphate Ca3(PO4)2, thereby releasing soluble phosphate ion in soils even at neutral pH, for uptake by plant life.” I would be very interested to see the evidence – but I cannot find anything in the scientific literature to support Sauerheber’s statement. It appears to be a figment of his imagination and anti-fluoride bias.
In the same unsupported manner, Sauerheber is suggesting silica (“orthosilicic acid”) may be dissolving our teeth. He even provides a chemical equation for it:
2H4SiO4 + Ca3(PO4)2 → 2HPO4-2 + 2H3SiO4– + 3Ca2+
First, the primary mineral in teeth is a bioapatite (Ca10(PO4)6(OH,F,Cl)2) not Ca3(PO4)2. And H3SiO4– is not stable at the pH of drinking water or saliva so his idea is destroyed by the immediate reaction:
H+ + H3SiO4–→ H4SiO4
In other words, Sauerheber’s equation above is driven to the left at the neutral and acid pH values of saliva and drinking water.
Incidentally, it is the presence of Ca2+, H2PO4– and F– in our saliva (derived from food and drink) that drives the dissolution equation for apatite to the left. It prevents dissolution (acid attack or demineralisation) and promotes remineralisation. This the surface or “topical” mechanism that reduces decay in existing teeth when fluoridated water is used.
Sauerheber’s confusion
Sauerheber’s arguments are chemically confused – probably because he is driven by a wish to find anything connected with fluoride to be bad. He is confused by terminology because the silica in solution is often called orthosilicic acid, or silicic acid. But the point is that this species (whatever it is – the chemistry of silica in water is very complex) is not dissociated at neutral pH values near 7. (more correctly only 0.18% of it is – Belton et al., 2012). It is a very weak acid -significant dissociation to form the anion only occurs at higher pH values according to the equation:
OH– + H4SiO4 → H3SiO4– + H2O
Enamel attack is caused by acid (H+) not an unionized silica species or silicate anion. At these high pH values, dissociation of silicic acid at high pH does not produce H+. It actually removes OH–.
The same confusion is behind Sauerheber’s assertion that leaching of lead from pipes and plumbing is caused by “orthosilicic acid.” He says:
“it is the intact orthosilicic acid, the predominant form present over the pH range 7-10 (sic) that is leaching lead or lead salts from pipes and plumbing fixtures.”
In fact, acid in drinking water is one of the causes of lead leaching. The chemical species responsible is H+ and that is why treatment plants adjust pH levels to reduce acidity. Silica in solution does not make a contribution to the (H+) concentration.
But if it did then we should be concerned about all water as fluoridating chemicals make only a minuscule contribution to silica in water.